Intro
For clinicians, “omega fats” are three distinct families with different biology, food sources, and clinical applications. Omega‑3 (n‑3) and omega‑6 (n‑6) are essential because humans cannot introduce double bonds beyond the ninth carbon, so their parent fatty acids, alpha‑linolenic acid (ALA, 18:3n‑3) and linoleic acid (LA, 18:2n‑6) must come from diet. Omega‑9 (n‑9), chiefly oleic acid (18:1n‑9), is monounsaturated and not essential, but has important cardiometabolic roles, especially when it replaces saturated fat. [1], [2], [3]
Omega 3 6 9 — what clinicians need to know at a glance
- Families & parents
- Omega‑3: ALA → EPA/DHA via desaturation/elongation; conversion is limited. [1], [5]
- Omega‑6: LA → γ‑linolenic acid (GLA) → dihomo‑γ‑linolenic acid (DGLA) → arachidonic acid (AA). [2], [5]
- Omega‑9: oleic acid is nonessential and abundant in olive oil, nuts, seeds, avocado.
- Enzyme competition
- ALA and LA compete for delta‑6 desaturase; high LA can reduce ALA conversion to EPA/DHA, though the clinical significance depends on overall intakes. [4], [5]
- Deficiency signal
- When dietary LA/ALA are insufficient, humans synthesize Mead acid (20:3n‑9). A rising triene:tetraene ratio (Mead/AA) indicates essential fatty acid deficiency. [6], [7]
- “Ratio” vs amounts
- Multiple authorities find little rationale for targeting a specific n‑6:n‑3 ratio if each is within recommended intakes; focus on adequate n‑3 and appropriate total PUFA. [8]
Biochemistry & metabolism clinicians counsel with
- Why LA and ALA are “essential”
- Humans lack delta‑12 and delta‑15 desaturases, so we cannot make LA or ALA (and cannot turn omega‑9 into omega‑6 or omega‑3). [3]
- ALA→EPA→DHA conversion is modest; reported population averages typically <10% to EPA and ≤1–4% to DHA. This underpins “food‑first for EPA/DHA” counseling. [1], [5]
- Pathways in brief
- Shared desaturases and elongases (FADS/ELOVL) process both n‑3 and n‑6 families; substrate competition occurs at delta‑6. [4], [5]
- With inadequate n‑3/n‑6 supply, oleic acid can be elongated/desaturated to Mead acid, a biomarker of essential fatty acid deficiency. [6], [7]
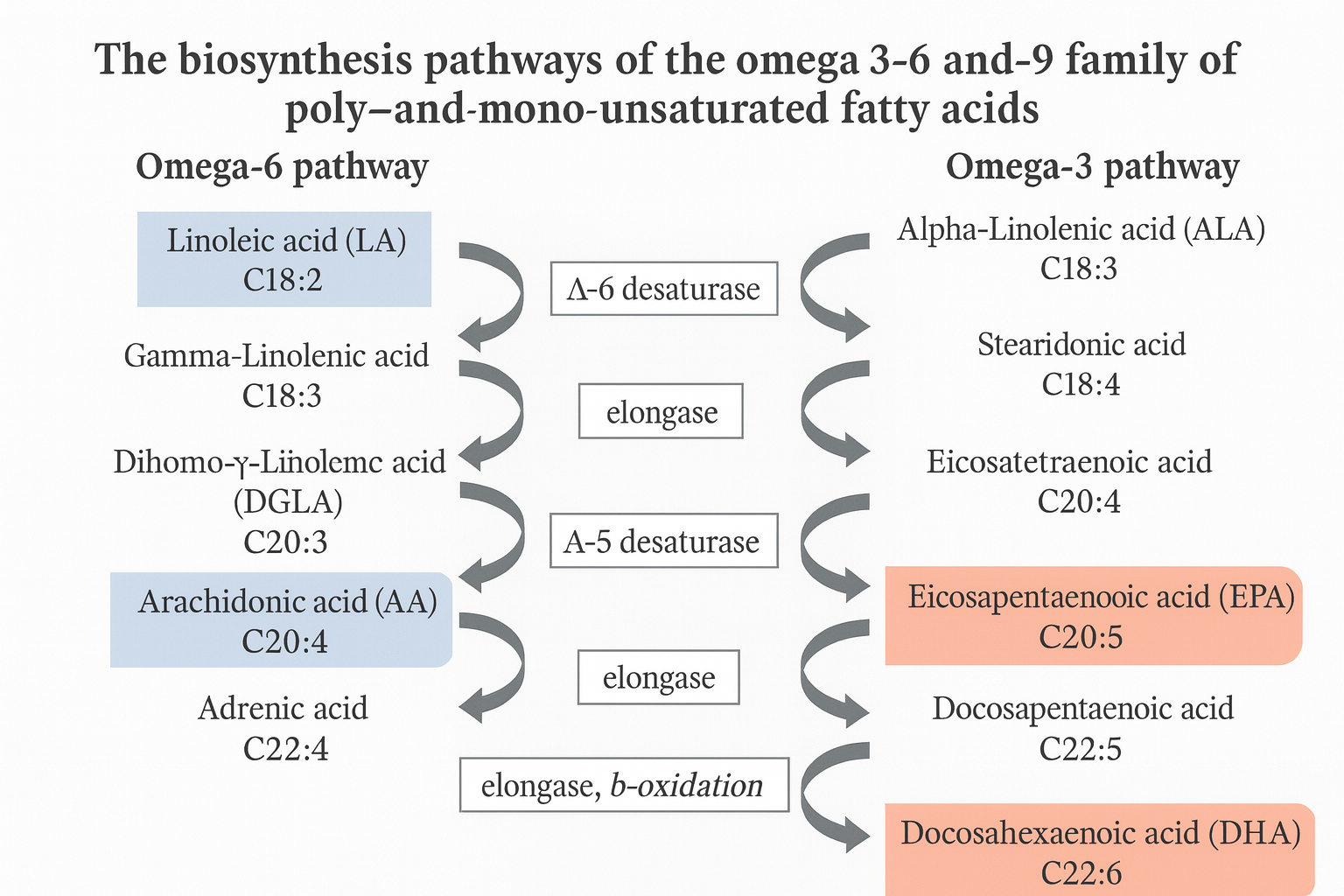
Biochemical pathways (Δ‑desaturases and elongases) — clinician detail
Key enzymes. FADS2 (Δ6‑desaturase), FADS1 (Δ5‑desaturase), and elongases (ELOVL5/ELOVL2). DHA completion uses the peroxisomal “Sprecher pathway.” [3–4]
n‑3 (omega‑3) pathway
- ALA (18:3n‑3) —Δ6 (FADS2)→ stearidonic acid (18:4n‑3) —ELOVL5→ ETA (20:4n‑3) —Δ5 (FADS1)→ EPA (20:5n‑3) —ELOVL2/5→ DPA (22:5n‑3) -Δ6 + peroxisomal β‑oxidation→ DHA (22:6n‑3).
- Notes: Endogenous ALA→EPA is modest; ALA→DHA is limited and diet/sex dependent. High LA intake can further reduce ALA conversion by competing at Δ6. [3,6–8]
n‑6 (omega‑6) pathway
- LA (18:2n‑6) —Δ6 (FADS2)→ GLA (18:3n‑6) -ELOVL5→ DGLA (20:3n‑6) -Δ5 (FADS1)→ AA (20:4n‑6).
- Notes: n‑6 and n‑3 families share Δ6; substrate competition is the bottleneck. [3–4,8–9]
n‑9 (omega‑9) in EFAD
- Oleic acid (18:1n‑9) -Δ6→ 18:2n‑9 -ELOVL5→ 20:2n‑9 -Δ5→ Mead acid (20:3n‑9).
- Notes: This is a deficiency workaround; it does not produce omega‑6. A higher triene:tetraene (Mead:AA) ratio indicates essential fatty acid deficiency. [3–5]
Mediator mapping (what these become)
- AA (n‑6) → 2‑series PG/TX and 4‑series LT; DGLA (n‑6) → 1‑series PG.
- EPA (n‑3) → 3‑series PG/TX and 5‑series LT; EPA/DHA → specialized pro‑resolving mediators (resolvins, protectins, maresins) that actively resolve inflammation. [26–27]
Structures & shorthand (for quick reference)
| Family | Common name | Shorthand (C:D n‑X) | Clinical role (very brief) | Primary sources |
|---|---|---|---|---|
| n‑3 | ALA | 18:3 n‑3 | Parent n‑3; limited EPA/DHA precursor | Flax, chia, walnuts, canola/soy |
| n‑3 | EPA | 20:5 n‑3 | TG lowering; eicosanoid/SPM substrate | Fatty fish, algal oils |
| n‑3 | DHA | 22:6 n‑3 | Neural/retinal membranes; SPMs | Fatty fish, algal DHA |
| n‑6 | LA | 18:2 n‑6 | Essential; skin barrier/ceramides; LDL‑C lowering when replacing SFA | Soy, corn, safflower, sunflower; nuts/seeds |
| n‑6 | GLA | 18:3 n‑6 | DGLA precursor; mixed supplement data | Evening primrose, borage |
| n‑6 | DGLA | 20:3 n‑6 | 1‑series PG substrate | Endogenous from LA/GLA |
| n‑6 | AA | 20:4 n‑6 | Eicosanoids; immune/vascular signaling | Meat, poultry, eggs |
| n‑9 | Oleic acid | 18:1 n‑9 | Cardiometabolic benefit when replacing SFA | EVOO, avocado, almonds |
| n‑9 | Mead acid | 20:3 n‑9 | EFAD marker (triene) | Endogenous in EFAD |
Citations for the pathways above are already present in your draft (enzymes/competition/conversion limits: [3–5,8–9]; SPM biology: [26–27]). No new sources needed.
Food sources & intake targets (U.S. practice)
- Foundational targets
- Adequate Intake (AI) from DRIs: LA (men 17 g/day, women 12 g/day; slightly lower ≥51y), ALA (men 1.6 g/day, women 1.1 g/day). [11]
- Pattern guidance: replace saturated fats with unsaturated fats (PUFA > MUFA) for CVD risk reduction. [9], [17]
- Omega‑3 foods
- EPA/DHA: fatty fish (salmon, sardines, herring, mackerel, anchovies, trout). The AHA recommends ~2 servings/week of fish, preferably fatty fish. [10], [17]
- ALA: flax, chia, walnuts, canola/soy oils; emphasize that ALA conversion to DHA is limited, and direct EPA/DHA intake strengthens status. [1]
- Plant‑based: algal‑oil DHA reliably raises DHA status in omnivores and vegans. [32]
- Omega‑6 foods
- LA: soybean, safflower, sunflower, corn oils; nuts and seeds. Clinical message: LA lowers LDL‑C when replacing saturated fat and is not inherently “pro‑inflammatory” in human data. [9], [26], [27]
- Omega‑9 foods
- Oleic acid: extra‑virgin olive oil, avocado, almonds, peanuts; typically cardioprotective when substituting for saturated fat, and prominent in the Mediterranean dietary pattern. [9]
- Practical fish guidance for sensitive populations
- FDA/EPA: 2–3 servings per week of “Best Choices” fish for pregnancy/lactation; emphasize low‑mercury options. [18]
What these fats do biologically
- Membranes & signaling
- Long‑chain PUFAs (AA, EPA/DHA) are membrane constituents that influence fluidity, raft organization, and cell signaling. [2], [5]
- Eicosanoids & resolution
- AA yields both pro‑ and anti‑inflammatory eicosanoids; DGLA yields series‑1 prostaglandins (less pro‑inflammatory). EPA competes with AA for eicosanoid synthesis and forms less potent analogs. [2], [5]
- Specialized pro‑resolving mediators (SPMs)-resolvins, protectins, maresins, are derived from EPA/DHA and actively promote resolution of inflammation and repair. [24], [25]
- Skin barrier
- LA is critical for epidermal ceramide synthesis and barrier integrity; EFAD leads to dermatitis and increased TEWL, reversible with LA repletion/topical approaches. [21], [22], [23]
Clinical evidence & indications — the quick map
Omega‑3 (EPA/DHA)
- Hypertriglyceridemia
- Prescription omega‑3 (EPA+DHA or EPA‑only) at 4 g/day lowers triglycerides and is recommended for severe hypertriglyceridemia; watch for LDL‑C rise with DHA‑containing products. [12]
- ASCVD outcomes
- Icosapent ethyl (pure EPA) 2 g BID reduced ischemic events and CV death in REDUCE‑IT (statin‑treated, elevated TG). [13]
- In contrast, STRENGTH (EPA+DHA carboxylic acids, 4 g/day) was neutral on MACE; formulation, placebo choice, and achieved EPA levels likely matter. [14]
- Safety signals
- Bleeding risk was not increased perioperatively in a randomized trial/meta‑analysis of fish oil use. [16]
- Dose‑related increase in atrial fibrillation risk has emerged, particularly at >1 g/day in outcome trials; discuss AF history and monitoring. [15]
- Counseling
- Food‑first (2 fish servings/week) for general prevention; reserve prescription therapy for hypertriglyceridemia or select high‑risk secondary prevention scenarios. [10], [17]
Omega‑6 (LA → AA)
- Lipids & CVD risk
- Replacing saturated fat with PUFA (including LA) lowers LDL‑C and reduces CHD events—supported by mechanistic and outcome evidence. [9], [26]
- Observational and intervention data show neutral or beneficial effects of n‑6 PUFA on cardiometabolic outcomes; human evidence does not support labeling LA as pro‑inflammatory. [27]
- Skin & mucosa
- LA is essential for epidermal barrier; deficiency causes dermatitis and elevated TEWL; correcting LA (diet or targeted topical lipids) supports barrier repair. [21], [22], [23]
- Supplements
- GLA‑rich oils (evening primrose, borage, black currant) do not improve eczema vs placebo in meta‑analysis; RA symptom relief shows mixed/modest evidence. [28], [29]
Omega‑9 (oleic acid)
- Cardiometabolic health
- Benefits are most evident when oleic acid replaces saturated fat within a Mediterranean pattern. PREDIMED (re‑analyzed) showed lower major CV events with extra‑virgin olive oil or nuts. [18]
- U.S. cohorts link higher olive oil intake with lower CHD/CVD risk. [19]
- For glycemia, controlled feeding trials favor PUFA over MUFA for improving insulin‑related endpoints, but MUFA‑rich patterns remain acceptable alternatives to SFA‑heavy diets. [20]
Myths & misconceptions clinicians often meet
- “Can omega‑9 convert to omega‑6?”
- No. Humans lack delta‑12 desaturase (and delta‑15), so we cannot insert the necessary double bonds to form LA or ALA de novo. Omega‑9 (oleic acid) cannot be turned into omega‑6 in humans. Under EFAD, oleic acid is diverted to Mead acid (20:3n‑9) instead. [3], [6], [7]
- “Should patients restrict omega‑6 to curb inflammation?”
- Routine restriction of LA is not evidence‑based. Major reviews and advisories emphasize adequate omega‑3 intake and replacement of saturated fat with unsaturated fats; specific n‑6:n‑3 ratio targets are not recommended when each class meets intake guidance. [8], [9], [26], [27]

Dosing & counseling — fast, practical algorithms
- General prevention
- Diet: 2 servings/week of fatty fish; use plant ALA daily; replace SFA with PUFA/MUFA. [10], [17]
- Intakes: aim to meet AI-LA 17 g/day (men) & 12 g/day (women); ALA 1.6 g/day (men) & 1.1 g/day (women). [11]
- Hypertriglyceridemia
- Consider prescription omega‑3 at 4 g/day (EPA‑only or EPA+DHA) for severe HTG; monitor LDL‑C if DHA‑containing. [12]
- Secondary prevention (ASCVD with elevated TG on statin)
- Discuss icosapent ethyl 2 g BID (outcome benefit), AF risk profile, and individual risk reduction goals. [13], [15]
- Skin barrier concerns
- Ensure adequate LA intake via mixed unsaturated oils/nuts/seeds; consider barrier‑repair topicals in AD. [21], [22], [23]
- Plant‑based patients
- Encourage ALA sources daily; consider algal‑oil DHA to improve status. [1], [32]
Special populations & safety
- Pregnancy & lactation
- FDA/EPA: choose low‑mercury “Best Choices” fish 2–3 times/week. [18]
- Cochrane suggests omega‑3 LCPUFA during pregnancy reduces preterm birth, with strongest signals for early preterm birth; evolving literature continues to refine dose/benefit. [31], [32]
- Algal DHA (e.g., 200–300 mg/day) is a practical option when fish is limited; supplemental DHA raises serum DHA in vegans and omnivores. [32]
- Anticoagulation & procedures
- High‑dose omega‑3s have not shown clinically significant perioperative bleeding increases; coordinate with proceduralists and continue individualized risk assessments. [16]
- Atrial fibrillation
- Briefly screen for AF history; discuss dose‑related AF signal seen in outcome trials using high doses of omega‑3; balance benefits/risks and monitor as indicated. [15]
Implementation pearls for clinics
- Write food first on the plan
- Translate targets into foods: “SMASH” fish (salmon, mackerel, anchovies, sardines, herring), nuts/seeds (walnuts, chia, flax), and liquid oils (soy, canola, safflower, EVOO). [10], [11]
- Focus on swaps
- Replace butter/tallow and high‑SFA snacks with EVOO, nuts, seeds, and fish to improve lipid risk factors and dietary quality. [9], [18], [19]
- Measure what matters
- For lipid management: TGs, LDL‑C (note DHA‑containing effects), non‑HDL‑C. For EFAD suspicion: consider triene:tetraene when clinically indicated. [12], [7]
- Communicate nuance
- Don’t demonize LA; ensure adequate n‑3 and appropriate total unsaturated fats. Emphasize patterns (Mediterranean‑style) over isolated nutrients. [9], [26], [27]
Key Takeaways
- Omega‑3 and omega‑6 are essential; omega‑9 is beneficial but nonessential. Humans cannot convert omega‑9 to omega‑6. [1], [3]
- EPA/DHA have clear roles in HTG lowering; EPA‑only therapy reduced events in REDUCE‑IT, while STRENGTH (EPA+DHA) was neutral. [12], [13], [14]
- LA is not inherently pro‑inflammatory in human data and supports skin barrier; restricting n‑6 is generally unnecessary when n‑3 intake is adequate. [21], [22], [23], [26], [27]
- Use food‑first strategies; consider algal DHA for plant‑based patients and prescription omega‑3 for lipid indications; discuss AF signal at high doses. [10], [12], [13], [15], [32]

References
Core biochemistry & physiology
[1] Omega-3 Fatty Acids – Health Professional Fact Sheet,” NIH Office of Dietary Supplements, updated 2022–2024.
[2] “Essential Fatty Acids,” Linus Pauling Institute, Oregon State University, accessed 2025.
[3] Lee J.M. et al., “Fatty Acid Desaturases, Polyunsaturated Fatty Acids, and Metabolic Disease,” Nutrients (2016) [PDF].
[4] Emery J.A. et al., “Δ-6 Desaturase Substrate Competition,” Int J Mol Sci (2013).
[5] Zhang J.Y. et al., “Desaturase and elongase limiting endogenous long chain PUFA synthesis,” Prog Lipid Res (2016).
[6] Kawashima H., “Physiological and pathological properties of Mead acid,” Int J Mol Sci (2023).
[7] Mogensen K.M., Parrish C.R., “Essential Fatty Acid Deficiency in Parenteral Nutrition,” Pract Gastroenterol (2017) [PDF].
[8] “Fats and Fatty Acids in Human Nutrition: Report of an expert consultation,” FAO/WHO (2010) [PDF].
Guidance & intake
[9] Sacks F.M. et al., “Dietary Fats and Cardiovascular Disease: A Presidential Advisory,” Circulation (2017).
[10] “Fish and Omega‑3 Fatty Acids,” American Heart Association, Aug 23, 2024.
[11] Institute of Medicine, “Dietary Reference Intakes for Macronutrients” (2002/2005) [PDF].
[17] Lichtenstein A.H. et al., “2021 Dietary Guidance to Improve Cardiovascular Health,” Circulation (2021).
[18] “Advice about Eating Fish,” U.S. FDA/EPA, Mar 5, 2024.
Omega‑3 clinical evidence & safety
[12] Skulas-Ray A.C. et al., “Omega‑3 Fatty Acids for the Management of Hypertriglyceridemia: A Science Advisory,” AHA (2019).
[13] Bhatt D.L. et al., “REDUCE‑IT,” N Engl J Med (2019).
[14] Nicholls S.J. et al., “STRENGTH,” JAMA (2020).
[15] Gencer B. et al., “Effect of Long‑Term Marine ω‑3 Fatty Acids on Atrial Fibrillation,” Circulation (2021).
[16] Akintoye E. et al., “Fish Oil and Perioperative Bleeding,” Circ Cardiovasc Qual Outcomes (2018).
Dietary patterns, omega‑9, and cardiometabolic outcomes
[18] Estruch R. et al., “Mediterranean Diet with EVOO or Nuts (PREDIMED reanalysis),” N Engl J Med (2018).
[19] Guasch‑Ferré M. et al., “Olive Oil Consumption and Cardiovascular Risk in U.S. Adults,” JACC (2020).
[20] Imamura F. et al., “Effects of SFA, PUFA, MUFA on Glucose‑Insulin Homeostasis,” PLoS Med (2016).
Omega‑6 physiology & inflammation; skin barrier
[26] Harris W.S. et al., “Omega‑6 Fatty Acids and Risk for Cardiovascular Disease,” Circulation (2009).
[27] Maki K.C. et al., “ω‑6 PUFAs and Cardiometabolic Health,” Nutrients (2018).
[21] Essential Fatty Acids and Skin Health,” Linus Pauling Institute, accessed 2025.
[22] Elias P.M. et al., “Evidence for a direct role for linoleic acid in barrier function,” J Invest Dermatol (1980).
[23] Wang X. et al., “The Role of Linoleic Acid in Skin and Hair Health: A Review,” Int J Mol Sci (2024).
Resolution biology & pro‑resolving mediators
[24] Ferreira I. et al., “Resolvins, Protectins, and Maresins: DHA‑Derived Mediators,” Int J Mol Sci (2022).
[25] Serhan C.N. et al., “Expert consensus: lipid mediators and inflammation resolution,” FASEB J (2024).
Eczema, RA, plant‑based strategies, and food composition
[28] Bamford J.T.M. et al., “Oral evening primrose oil and borage oil for eczema,” Cochrane (2013).
[29] Cochrane, “Herbal therapy for rheumatoid arthritis” (GLA oils), updated 2022.
[32] García‑Maldonado E. et al., “Algal‑oil DHA increases serum DHA in omnivores and vegans,” Nutrients (2022).
[34] USDA National Nutrient Database, “DHA content of foods,” Release 28 (2016) [PDF].
Pregnancy & preterm birth
[31] Middleton P. et al., “Omega‑3 LCPUFA addition during pregnancy,” Cochrane Review (2018).
[32] Fu J.Y.X. et al., “Role of omega‑3 PUFA in prevention of preterm birth,” Med J Aust (2024).
