Why the liver’s clearance system matters to whole‑body health
The liver receives ~25% of cardiac output and is uniquely positioned between the gut and the systemic circulation. Its lobular architecture brings xenobiotics, dietary constituents, hormones, and microbial metabolites into intimate contact with hepatocytes and non-parenchymal cells. Hepatic “detox” is not a single pathway but an orchestrated network of (i) uptake transporters, (ii) activating and inactivating enzymes, and (iii) efflux pumps that deliver water‑soluble products to bile or urine. At steady state, this network protects against both external exposures (e.g., drugs, pollutants) and internal byproducts (e.g., bilirubin, bile acids, lipid peroxidation aldehydes) while minimizing collateral oxidative stress [1–3].
A practical way to teach patients is to emphasize conversion plus clearance: most fat‑soluble molecules must be chemically altered (Phase I), attached to a polar handle (Phase II), and exported (Phase III). The system is plastic dietary patterns, body composition, inflammation, circadian rhythms, and genetics can upregulate or constrain capacity [4,5].
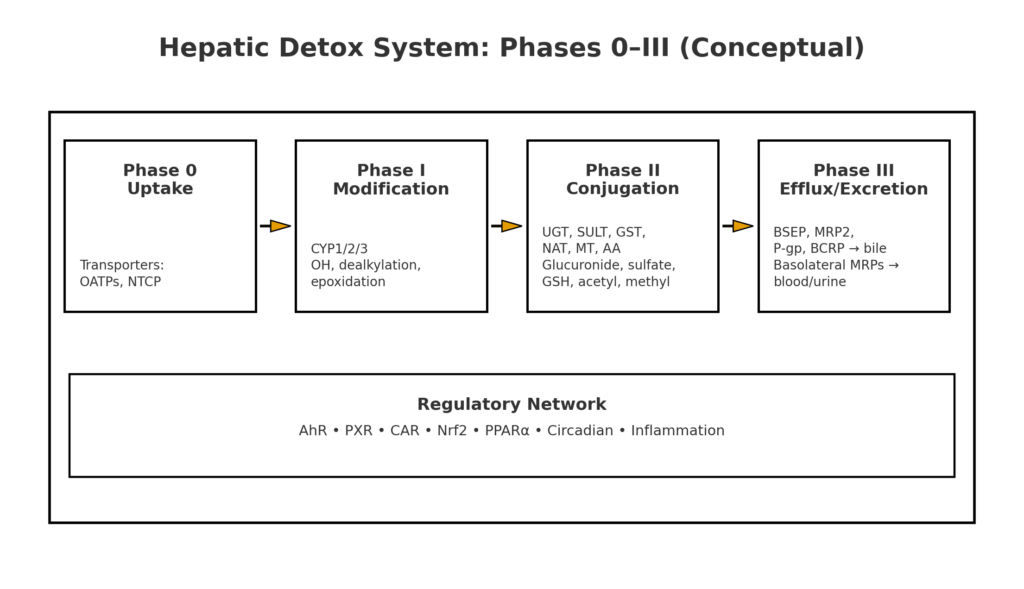
Table of Contents
Mechanistic overview of liver detoxification pathways
Phase 0 (uptake). Basolateral transporters move substrates from portal blood into hepatocytes: NTCP for bile acids; OATPs (SLCO1B1/1B3) for many drugs, hormones, and bilirubin conjugates [6,7].
Phase I (activation/modification). Cytochrome P450s (mainly families 1, 2, 3) perform oxidations such as aliphatic and aromatic hydroxylation, N‑, O‑ or S‑dealkylation, epoxidation, sulfoxidation, and more. The same chemistry that increases polarity can also generate reactive intermediates, hence the need for robust Phase II and antioxidant defenses [1–3]. Representative reactions include:
- Hydroxylation: RH → ROH (CYP3A4, CYP2D6, CYP2C19) [1,2]
- Dealkylation: R–N(CH₃)₂ → R–NHCH₃ + CH₂O (CYP2D6/3A) [3]
- Epoxidation: polycyclic aromatics → arene oxides (CYP1A1/1B1) [2]
Transcriptional control. Phase I/II/III genes are coregulated by xenobiotic sensors-AhR, PXR, CAR, and by stress-response factors like Nrf2. These receptors integrate drug exposures, bile acids, fatty acids, and inflammatory signals to tune enzyme abundance and transporter expression [4,5].
Phase II (conjugation). Polar “handles” are attached to reactive intermediates to neutralize and mobilize them:
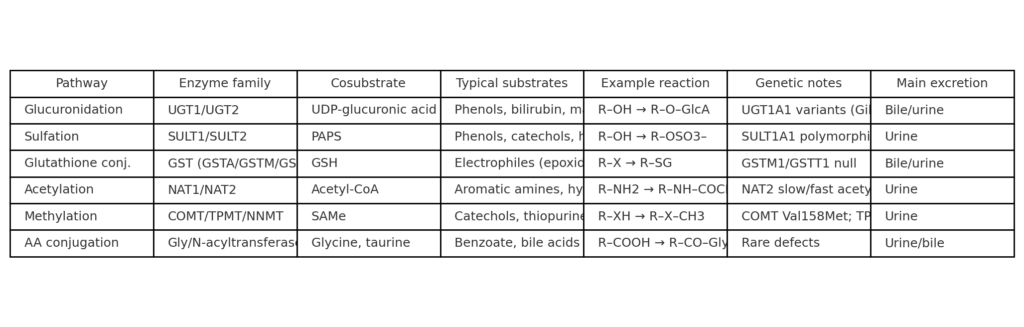
- Glucuronidation (UGTs): the most prevalent conjugation across drugs and endobiotics (e.g., bilirubin, steroid hormones) [1]. UDP‑glucuronic acid is the co‑substrate; UGT1A1 is crucial for bilirubin [13].
- Sulfation (SULTs): uses PAPS to sulfate phenols, catechols, and small amines; high capacity in youth, more easily saturated at typical adult exposures [9].
- Glutathione conjugation (GSTs): attaches GSH to electrophiles (epoxides, quinones), preventing macromolecule adduction and facilitating export [10,11].
- Acetylation (NATs): detoxifies aromatic amines/hydrazines; polymorphic NAT2 yields “slow” and “fast” acetylators—clinically significant for isoniazid, among others.
- Methylation (COMT/TPMT/NNMT): SAM‑dependent methyl donors neutralize catechols (COMT) or thiopurines (TPMT); one‑carbon nutrition (folate, B12, choline, betaine) sustains SAMe pools [28–30].
- Amino acid conjugation: glycine and taurine conjugate carboxylates (e.g., benzoate→hippurate; bile acids→glyco/taurine conjugates), enhancing polarity and transport [25,26].
Phase III (efflux). Canalicular pumps (BSEP/ABCB11; MRP2/ABCC2; P‑gp/ABCB1; BCRP/ABCG2) export conjugates into bile; basolateral MRPs provide a “safety valve” back to blood for renal excretion. Transporter disruption genetic or drug‑induced can precipitate cholestatic liver injury [6–8,27].
From biochemistry to bedside: common clinical touchpoints
Acetaminophen (APAP). Therapeutic doses are primarily glucuronidated and sulfated; a minor CYP2E1‑mediated fraction forms NAPQI, which is detoxified by glutathione. Overdose or depleted GSH shifts flux toward hepatotoxicity. N‑acetylcysteine (NAC) replenishes cysteine/GSH and directly reduces NAPQI; it is most effective when started within 8 hours, guided by the Rumack–Matthew nomogram for single acute ingestions [23,24].
Bilirubin physiology. Unconjugated bilirubin is glucuronidated by UGT1A1. In Gilbert syndrome, reduced UGT1A1 activity produces benign, intermittent unconjugated hyperbilirubinemia often unmasked by fasting or illness. Selected drugs inhibit UGT1A1 and can raise bilirubin; this is a useful, real‑world illustration of how conjugation capacity modulates biomarker levels [13].
Transporter-mediated drug interactions. OATP1B1/1B3 uptake supports hepatic extraction; inhibition raises systemic exposure (e.g., statins). BSEP inhibitors may precipitate cholestasis; MRP2 impairment shifts conjugates to plasma via MRP3/4. These effects are now built into DDI decision trees used by developers and regulators [6–8,27].
Diet, lifestyle, and environmental context for U.S. and Illinois readers
Metabolic health and MASLD. Weight reduction (approximately 7-10%) improves steatosis and inflammation; consensus guidelines emphasize Mediterranean‑style eating patterns, regular physical activity, and management of cardiometabolic risks. Coffee consumption (2-4 cups/day of caffeinated coffee, without excess sugar) is consistently associated with lower fibrosis risk and improved liver outcomes in MASLD cohorts; clinicians can reasonably encourage coffee in appropriate adults without contraindications [17–19].
Illinois‑specific exposure notes.
• Sport fish consumption advisories. The Illinois Department of Public Health updates species‑ and waterbody‑specific advisories for mercury, PCBs, and PFOS. Sensitive populations (children, people who are nursing, pregnant, or may become pregnant) should adhere to posted meal frequencies; statewide guidance limits predatory fish for sensitive groups to ~1 meal/week. Use the IDPH advisory tables/map before recommending local catch consumption [20].
• Metals and occupational risks. For suspected arsenic exposure, order a 24‑hour urinary arsenic with speciation (to distinguish toxic inorganic from seafood‑derived organic forms). For significant lead exposure, treatment decisions including chelation follow CDC/ATSDR guidance; cessation of exposure is paramount [21,22].
• Indoor/environmental hygiene. Ventilate living spaces, minimize smoke exposure, and follow safe food and water guidance. These simple steps reduce Phase I burden and downstream conjugation demand.

Nutrition & supplement evidence—what helps, what doesn’t
What is supported.
• Whole dietary patterns. Emphasize minimally processed foods, adequate protein, high fiber, and a diversity of plants; these patterns support one‑carbon metabolism (for methylation), cysteine availability (for glutathione), and glycine/taurine (for conjugation).
• Coffee. Moderate, unsweetened coffee intake correlates with less fibrosis and lower liver stiffness in population studies and cohort analyses [17–19].
• Cruciferous vegetables. Clinical trials in heavily polluted settings show broccoli sprout beverages increase urinary excretion of benzene and acrolein metabolites via glutathione‑dependent pathways, consistent with Nrf2‑mediated induction of cytoprotective enzymes [14,15]. Effects are context‑dependent; not all trials show pathway activation (e.g., a COPD Nrf2‑target gene study was negative) [16].
What to avoid.
• Detox cleanses and “liver flushes.” High‑quality evidence does not support commercial detox diets for toxin elimination or durable weight loss; case reports describe herbal “cleanse”–induced hepatitis. Focus patients on sustainable nutrition, hydration, sleep, and physical activity rather than short‑term cleanses [16].
Genetics, epigenetics, and circadian control: why patients respond differently
Interindividual variation arises from heritable and acquired factors:
• CYPs/UGTs/SULTs/GSTs. Allelic variation alters enzyme abundance/activity (e.g., GSTM1/GSTT1 null genotypes, SULT1A1 variants). UGT1A1 promoter polymorphisms in Gilbert syndrome reduce bilirubin glucuronidation [10,11,13].
• NAT2 acetylation phenotype. Clinically relevant for several aromatic amines/hydrazines.
• Methylation enzymes. COMT (catechols) and TPMT (thiopurines) influence drug response; TPMT/NUDT15 pharmacogenetics is established standard of care in thiopurine therapy [28,29].
• Regulatory networks. AhR, PXR, CAR, and Nrf2 coordinate gene expression in response to diet, xenobiotics, bile acids, and inflammatory signals; circadian clocks also tune detox capacity, which may partly explain time‑of‑day differences in drug handling [4,5].
Testing, monitoring, and red flags (clinician checklist)
- Medication/exposure inventory: acetaminophen totals, alcohol pattern, supplements/herbals, occupational and hobby exposures, local fish intake, vaping/smoke, solvents. Educate on label reading and dose stacking.
- Laboratory screening: ALT, AST, ALP, GGT, bilirubin fractionation, albumin, INR; lipid profile and A1c for metabolic risk; ferritin and transferrin saturation if indicated.
- Viral hepatitis: follow national guidance for hepatitis C screening (universal one‑time for adults; periodic for ongoing risk) and vaccination for hepatitis A/B per risk profile.
- When concerned about metals: 24‑hour urinary arsenic with speciation; BLL for lead; escalate per CDC/ATSDR thresholds [21,22].
- Drug‑specific tools: Rumack–Matthew nomogram with timely acetaminophen level for suspected overdose; initiate NAC promptly when indicated [23,24].
- Transporter/DDI awareness: statins (OATP1B1), BSEP‑inhibiting drugs in cholestasis, and P‑gp/BCRP interactions in polypharmacy [6–8,27].
Clinical checklist for strengthening hepatic clearance
- Dietary pattern: Mediterranean‑style, protein‑adequate (to supply cysteine, glycine, taurine), high in cruciferous vegetables and fiber‑rich foods; minimize ultra‑processed foods.
- Weight and movement: steady weight loss when indicated; combination of aerobic and resistance training for insulin sensitivity and hepatic fat reduction [17].
- Hydration and bile flow: maintain fluid intake; dietary fiber supports enterohepatic bile acid cycling and fecal toxin removal.
- Coffee: 2–4 cups/day (if tolerated and without contraindications) is reasonable for adults with MASLD risk [17–19].
- Alcohol: adhere to national guidance and individualize; for steatotic liver disease, advise no or minimal alcohol.
- Medication stewardship: avoid unnecessary polypharmacy; prefer agents with safer hepatic profiles when choices exist.
- Environmental hygiene (Illinois): check IDPH fish advisories before eating local catch; address workplace exposures promptly [20–22].
Key Takeaways
- Hepatic clearance is a network-uptake (Phase 0), modification (Phase I), conjugation (Phase II), and efflux (Phase III)-regulated by nutrient, hormonal, and xenobiotic signals [1,4–8].
- Phase I creates more polar (sometimes reactive) intermediates; Phase II neutralizes them via glucuronidation, sulfation, glutathione, acetylation, methylation, and amino acid conjugation [1–3,9–11,25,26].
- Real‑world cases: acetaminophen overdose, Gilbert syndrome, transporter DDIs illustrate how pathway capacity limits clinical outcomes [6,13,23,24,27].
- Lifestyle interventions (diet quality, weight loss, physical activity, coffee, targeted crucifer intake) align with best evidence, whereas detox cleanses lack supportive data [14–19].
- Illinois readers should use IDPH fish advisories and CDC/ATSDR guidance for metals; targeted testing and exposure cessation are more effective than unproven “detoxes” [20–22].

References
Core physiology and Phase I enzymes
[1] Zanger & Schwab. “Cytochrome P450 enzymes in drug metabolism: regulation and function.” Pharmacol Ther. 2013. PubMed
[2] Guengerich FP. “Mechanisms of cytochrome P450–catalyzed oxidations.” ACS. 2007. PubMed
[3] Isin EM, Guengerich FP. “Substrate binding to cytochromes P450.” Curr Drug Metab. 2007. ScienceDirect
Transcriptional regulation and stress responses
[4] Aleksunes LM et al. “Coordinated regulation of hepatic Phase I and II drug-metabolizing genes and transporters by AhR, CAR, PXR, PPARα, and Nrf2.” Drug Metab Dispos. 2012. PMC
[5] Zhou J et al. “The Nrf2 pathway in liver diseases.” Front Cell Dev Biol. 2022. Frontiers
Transporters and Phase III efflux
[6] Taskar KS et al. “Clinical relevance of hepatic and renal P‑gp/BCRP inhibition: ITC perspective.” Clin Pharmacol Ther. 2022. PMC
[7] Tátrai P et al. “Role of hepatocyte transporters in drug‑induced liver injury.” Biomedicines. 2022. PMC
[8] Mohammed TA et al. “The role of hepatobiliary transporters in bile acid homeostasis.” Egypt Liver J. 2025. SpringerOpen
[27] Järvinen E et al. “Uptake and efflux transporters in the liver interplay and disease modulation.” Front Pharmacol. 2022. Frontiers
Phase II conjugation (selected)
[9] Xie Y et al. “The role of sulfotransferases in liver diseases.” Drug Des Devel Ther. 2020. PMC
[10] Hayes JD et al. “Glutathione transferases.” Annu Rev Pharmacol Toxicol. 2005. Europe PMC
[11] Alnasser SM et al. “The role of glutathione S‑transferases in human disease.” Int J Mol Sci. 2024. PMC
[12] Cook I et al. “SULT1A1 substrate selectivity.” Drug Metab Dispos. 2015. PMC
[25] Reactome Pathway: “Conjugation of benzoate with glycine.” Reactome
[26] Shulpekova Y et al. “Bile acid metabolism and conjugation ratio.” Int J Mol Sci. 2022. PMC
Endogenous exemplars
[13] Vítek L. “Gilbert’s syndrome revisited.” J Hepatol. 2023. Journal of Hepatology
Lifestyle and clinical trials
[14] Egner PA et al. “Broccoli sprout beverage increases detoxication of airborne pollutants.” Cancer Prev Res. 2014. PMC
[15] Chen JG et al. “Dose‑dependent detoxication of benzene with broccoli sprout beverage.” Am J Clin Nutr. 2019. PubMed
[16] Wise RA et al. “Lack of effect of oral sulforaphane on Nrf2 target gene expression in COPD.” PLoS One. 2016. PLOS
[17] AASLD Practice Guidance: Metabolic dysfunction‑associated steatotic liver disease. Hepatology. 2023. PMC
[18] AASLD announcement, 2025 MASLD update (semaglutide considerations). 2025. AASLD
[19] Di Pietrantonio D et al. “Protective effect of coffee constituents on liver disease: meta‑analysis.” Nutrients. 2024. PMC